Blog
Mammography image quality, News

12 Apr 2024
Volpara Launches MQSA Compliance Software at the Society for Breast Imaging Symposium
Breast cancer, Mammography image quality
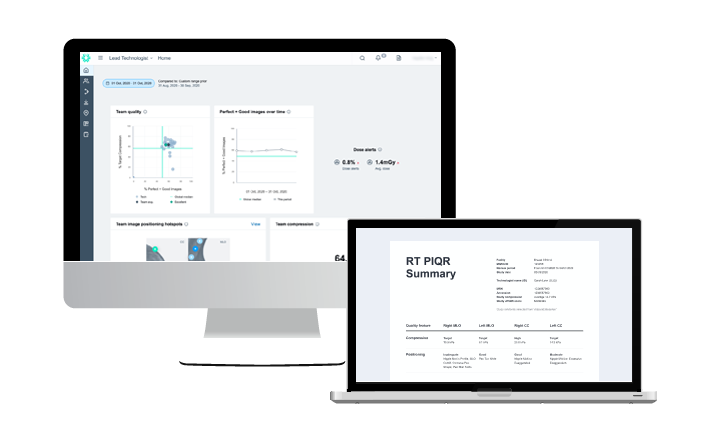
31 Mar 2024
AI to help your mammography MQSA audit…yes, really
News

15 Mar 2024
Volpara to Showcase AI Software for Breast Centers at NCBC 2024, Including Lunit INSIGHT for Early Detection
News

21 Feb 2024
Volpara to Highlight Growing AI Suite for Mammography at ECR 2024, Including New Lunit Distribution Agreement
News

20 Dec 2023
Volpara Health breast density and mammography quality software highlighted in new peer-reviewed research
News

27 Nov 2023
Volpara Health and LehYup Partner on Cancer Risk Assessment Services at RSNA 2023
Volumetric breast density

24 Oct 2023
AI that Works in Breast Imaging: Volpara literally wrote the eBook!




